Revolutionizing Brain-Computer Interaction: Neuralink Receives FDA Approval for Human Clinical Trials
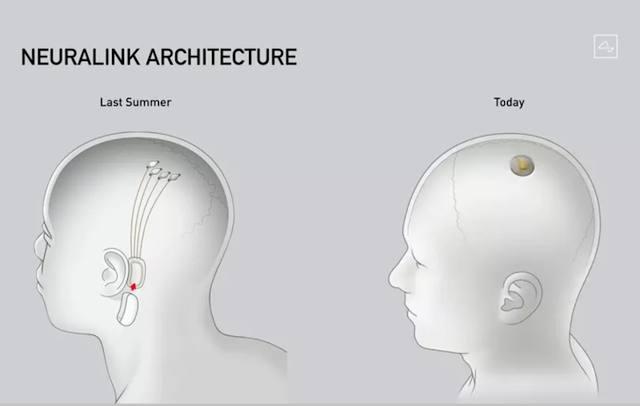 |
| "The difference between last year's Neuralink BMI chip, which was implanted near the ear (left), and the latest device aligned with the skull (right). (Neuralink via The Verge)" |
Neuralink, the brainchild of entrepreneur Elon Musk, has recently announced a major breakthrough in its quest to enable direct brain-computer interaction. The start-up, which aims to develop groundbreaking neurotechnology, has received approval from the US Food and Drug Administration (FDA) to conduct its first-ever clinical study involving human subjects. This significant milestone marks a crucial step forward for Neuralink's technological advancement.
Excitement fills the air as Neuralink took to Twitter, proclaiming, "We are excited to share that we have received the FDA's approval to launch our first-in-human clinical study! This is the result of incredible work by the Neuralink team in close collaboration with the FDA and represents an important first step that will one day allow our..." The tweet underscores the tremendous efforts made by the Neuralink team in close cooperation with the FDA, demonstrating their commitment to pioneering this groundbreaking technology.
Elon Musk, in his capacity as Neuralink's spokesperson, expressed his delight with the FDA's approval. He had previously articulated the purpose of Neuralink's brain implant—to enable direct communication between the human brain and computers. Musk stated, "We have been working diligently to prepare for our first human implant, and we want to be extremely cautious and ensure that it works well before introducing the device to humans," as quoted from a Neuralink presentation reported by AFP.
It is worth noting that Musk has been known for making ambitious predictions regarding his ventures, with some falling short of fruition. In July 2019, he vowed that Neuralink would conduct its first human tests in 2020. Demonstrations showcased Neuralink's prototype device, a coin-sized chip implanted in a monkey's skull, allowing it to interact with basic video games or move a cursor on a screen.
During the Neuralink presentation, the company exhibited how several monkeys were able to "play" basic video games or control a cursor through their Neuralink implants. This time, the chips will undergo trials by being implanted into the human brain. Musk revealed that the initial focus would be on utilizing the implant to restore vision and mobility for individuals who have lost these abilities. "Initially, we'll enable someone with almost no ability to move their limbs to operate a smartphone faster than someone using thumbs," he stated. "While it may sound miraculous, we believe restoring full-body functionality to someone with a severed spinal cord is possible," Musk explained.
 |
| "Tesla CEO Elon Musk donned a batik attire and kept a low profile while speaking at the B20 Summit Indonesia 2022. (YouTube/B20 Indonesia 2022)" |
Beyond the potential to treat neurological disorders, Neuralink aims to ensure that humans do not become intellectually overwhelmed by artificial intelligence. Another company, Synchron, has been working on a similar system and recently announced the successful implantation of the first brain-machine interface device in the United States in July.
The approval from the FDA paves the way for Neuralink's revolutionary technology to progress further. As the company embarks on its first human clinical trials, the world eagerly awaits the results and the potential implications for the future of brain-computer interaction. With Neuralink's groundbreaking endeavors, a new era of technological innovation may soon be upon us, transforming the way we perceive and interact with the world around us.